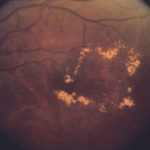
We are proud to announce that Global Clinical Trials (GCT) has gained EC and RA approval last week in Hungary for the ongoing ophthalmology project. This approval allows GCT to continue to demonstrate its well-established knowledge and experience in conducting clinical trials across various ophthalmic indications. The clinical sites in Hungary are part of a large multinational, phase II diabetic macular edema clinical study being conducted within the European Union.
GCT has performed numerous clinical trials in ophthalmology over the years. We maintain close relationships with KOLs who specialize in ophthalmology, as well as with high-recruiting investigative sites, and will gladly apply this knowledge across multiple therapeutic areas and indications to ensure the successful completion of your planned clinical program.
Please contact us at bd@gctrials.com for guidance and expertise on how best to navigate the clinical, regulatory and logistical aspects of conducting pharmaceutical and medical device trials in Hungary as well as other countries.