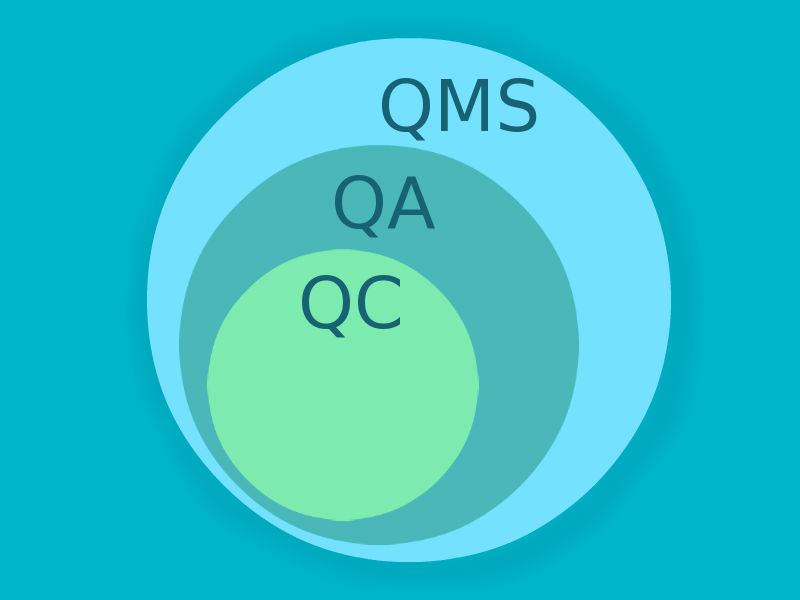
It is never enough when it comes to quality. GCT quality assurance system is meant to increase customer confidence and company’s credibility. Throughout the years we have passed over 65 internal and external audits with no major findings and are not going to slow down.
This month we are happy to welcome the accomplished pharma business professional as a new member of our dedicated Quality Assurance team in Russia.
GCT conducts independent vendor/facility audits in compliance with ICH guidelines and other applicable regulatory requirements. Our QA Department is in close contact with QA experts in the region and we are happy to engage professionals with a profound clinical research experience in our work. Our team of highly professional QA experts with 18+ years of experience in clinical research and a solid record of audits and inspections will ensure the integrity of your clinical trials.
To order an independent GCP audit for reasonable price please contact us by bd@gctrials.com